Competition between folded and extended structures of alanylalanine (Ala-Ala) in a molecular beam†
Abstract
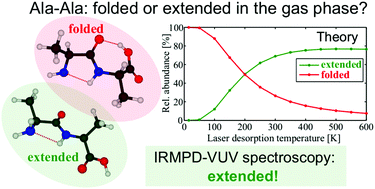
We report the gas phase conformational preferences of laser desorbed Ala-Ala dipeptides probed by action spectroscopy using the IRMPD-VUV method. The molecules were internally cooled through collisional cooling in a supersonic jet environment. An IR spectrum was obtained experimentally in the spectral range of 700–1850 cm−1, and subsequently interpreted with the help of quantum chemical calculations. Although theory predicts that folded structures have lower electronic energies and, thus, are more stable at low temperatures compared to their extended (β-strand-like) counterparts, analysis of the experimental data concluded the extended conformer to be the most dominant. An explanation to this observation is discussed in this paper and rationalized in terms of collisional conformer relaxation processes occurring in the supersonic jet molecular beam.
- This article is part of the themed collections: 2019 PCCP HOT Articles and Photodissociation and reaction dynamics


 Please wait while we load your content...
Please wait while we load your content...