P-NIPAM in water–acetone mixtures: experiments and simulations†
Abstract
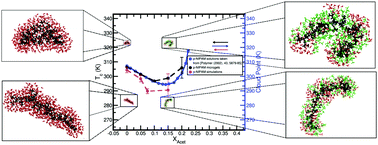
The lower critical solution temperature (LCST) of poly-N-isopropylacrylamide (p-NIPAM) diminishes when a small volume of acetone is added to the aqueous polymer solution, and then increases for further additions, producing a minimum at a certain acetone concentration. Here this behavior is observed through the variation of the hydrodynamic radius RH of p-NIPAM microgels with temperature, measured by dynamic light scattering (DLS), when adding increasing amounts of acetone in the molar fraction range of 0.00 to 0.25. This size trend of microgels with temperature is well captured by all-atom molecular dynamic simulations, which are implemented for a single 30-mer, at similar solvent and temperature conditions. Both DLS measurements and simulations indicate that the shrunken state continuously augments its size with increasing acetone content. This, in turn, leads to a minimum of the globule-to-coil transition temperature, which should correspond to the minimum of the LCST. Furthermore, density profiles, as obtained by considering a membrane arrangement of oligomers, reveal a preferential interaction of the polymer with acetone to the detriment of water. We observe how the membrane loses water content as the temperature is increased while keeping a similar amount of acetone in its interior. This competition between water and acetone for the polymer surface plays a major role in the enthalpy driven dependence of the critical temperature with acetone concentration.
- This article is part of the themed collection: Celebrating recent chemical science in Mexico


 Please wait while we load your content...
Please wait while we load your content...