Thermodynamic stability of non-stoichiometric SrFeO3−δ: a hybrid DFT study†
Abstract
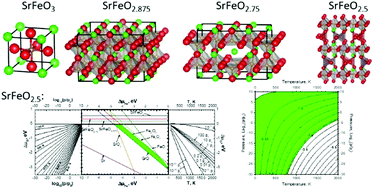
SrFeO3−δ is a mixed ionic-electronic conductor with a complex magnetic structure that reveals a colossal magnetoresistance effect. This material and its solid solutions are attractive for various spintronic, catalytic and electrochemical applications, including cathodes for solid oxide fuel cells and permeation membranes. Its properties strongly depend on oxygen non-stoichiometry. Ab initio hybrid functional approach was applied herein to study the thermodynamic stability of a series of SrFeO3−δ compositions with several non-stoichiometries δ, ranging from 0 to 0.5 (SrFeO3–SrFeO2.875–SrFeO2.75–SrFeO2.5) as a function of temperature and oxygen pressure. The results obtained by two approaches, in which either (i) all electrons at Fe atoms explicitly described or (ii) inner core electrons at Fe atoms are replaced by effective core potential, are compared. Based on our calculations, phase diagrams were constructed, allowing the determination of environmental conditions for the existence of stable phases. It is shown that (within an employed model) only the SrFeO2.5 phase appears to be stable. The stability region for this phase was re-drawn on the contour map of oxygen chemical potential, presented as a function of temperature and oxygen partial pressure. A similar analysis was also performed using experimental Gibbs energies of perovskite formation from the elements. The present modelling strongly suggest a significant attraction between neutral oxygen vacancies. These vacancies are created during a series of the abovementioned SrFeO3−δ mutual transformations accompanied by oxygen release.


 Please wait while we load your content...
Please wait while we load your content...