Water diffusion measurements of single charged aerosols using H2O/D2O isotope exchange and Raman spectroscopy in an electrodynamic balance†
Abstract
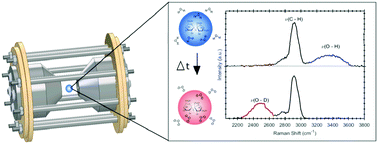
Sea spray aerosols contain a large array of organic compounds that contribute to high viscosities at low relative humidity and temperature thereby slowing translational diffusion of water. The Stokes–Einstein equation describes how viscosity is inversely correlated with the translational diffusion coefficient of the diffusing species. However, recent studies indicate that the Stokes–Einstein equation breaks down at high viscosities achieved in the particle phase (>1012 Pa s), underestimating the predicted water diffusion coefficient by orders of magnitude and revealing the need for directly studying the diffusion of water in single aerosols. A new method is reported for measuring the water diffusion coefficient in single suspended charged sucrose–water and citric acid (CA)–water microdroplets in the 30–60 micron diameter range. The translational water diffusion coefficient is quantified using the H2O/D2O isotope exchange technique between 26 and 54% relative humidity (RH) for sucrose and 7 and 25% RH for CA using a recently developed mobile electrodynamic balance apparatus. The results are in good agreement with the literature, particularly the Vignes-type parameterization from experiments using isotope exchange and optical tweezers. Below 15% RH, CA droplets show incomplete H2O/D2O exchange. This mobile electrodynamic balance will allow future studies of atmospherically relevant chemical systems, including field studies.


 Please wait while we load your content...
Please wait while we load your content...