Insights into the ion-exchange properties of Zn(ii)-incorporated MOR zeolites for the capture of multivalent cations†
Abstract
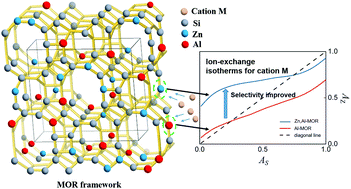
Understanding the properties of zeolites for cation exchange is important because the ion-exchange performance largely determines their suitability in applications such as catalysis and adsorptive separation. We synthesized a Zn(II)-incorporated mordenite-framework aluminosilicate zeolite (Zn,Al-MOR), in which both Zn and Al are substituted in the framework, and studied its ion-exchange behavior for multivalent cations. For comparison, the original aluminosilicate mordenite (Al-MOR) was also synthesized with a composition adjusted to ensure that its charge density was similar to that of Zn,Al-MOR. While the incorporation of Zn(II) led to a slower kinetic process, the selectivities and the exchange capacities toward multivalent cations (especially divalent cations) were significantly improved. Herein, we discussed the mechanism responsible for improving the ion-exchange performance in the presence of Zn(II) and found that the incorporation of Zn(II) led to a significant improvement in the ion-exchange temperature dependence of the MOR, which led to the ability to enhance ion-exchange capacity through temperature control during actual application. It was also revealed that the presence of Zn(II) significantly improves selectivity and spontaneity toward the exchange of multivalent cations, Ni2+. Moreover, XRD and nitrogen-adsorption/desorption analyses revealed that the structures of the materials are maintained during the ion exchange, which is indicative of superior structural stability and tolerance to ion exchange.


 Please wait while we load your content...
Please wait while we load your content...