Understanding CO oxidation on the Pt(111) surface based on a reaction route network†
Abstract
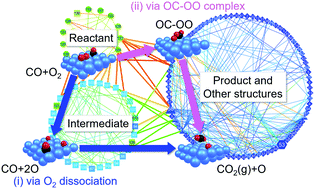
Analysis of a reaction on a solid surface is an important task for understanding the catalytic reaction mechanism. In this study, we studied CO oxidation on the Pt(111) surface by using the artificial force induced reaction (AFIR) method. A systematic reaction path search was done, and the reaction route network was created. This network included not only bond rearrangement paths but also migration paths of adsorbed species. Then, the obtained network was analyzed using a kinetics method called rate constant matrix contraction (RCMC). It is found that the bottleneck of the overall reaction is the CO2 generation step from an adsorbed CO molecule and an O atom. This result is consistent with the Langmuir–Hinshelwood (LH) mechanism with O2 dissociation discussed in previous studies. The present procedure, i.e., construction of the reaction route network by the AFIR method followed by application of the RCMC kinetics method to the resultant reaction route network, was fully systematic and uncovered two aspects: the impact of the existence of multiple paths in each bond rearrangement step and an entropic contribution arising from short-range migration of adsorbed species.
- This article is part of the themed collection: Photodissociation and reaction dynamics


 Please wait while we load your content...
Please wait while we load your content...