Photoelectron spectroscopic study of I−·ICF3: a frontside attack SN2 pre-reaction complex
Abstract
Photodetachment and 2D photoelectron spectra of the mass-selected I−·CF3I complex are presented together with electronic structure calculations. Calculations show that the I− is located at the iodine side of CF3I. Vertical and adiabatic detachment energies were measured at 4.03 and approximately 3.8 eV, respectively. The photoelectron spectra and molecular orbitals show a significant covalent bonding character in the cluster. The presence of electronic excited states is observed. Below threshold, iodide is generated which can be assigned to the photoexcitation of degenerate charge-transfer bands from the off-axis p-orbitals localised on iodide. Near the onset of two spin–orbit thresholds, bright excited states are seen in the experiment and calculations. Excitation of these leads to the formation of slow electrons. The spectroscopy of I−·CF3I is compared to the well-studied I−·CH3I cluster, a pre-reaction complex in the text-book I− + CH3I SN2 reaction. Despite the reversed stereodynamics (i.e. inversion of the CX3 between X = H and F) of the SN2 reaction, striking similarities are seen. Both complexes possess charge transfer excited states near their respective vertical detachment energies and exhibit vibrational structure in their photoelectron spectra. The strong binding is consistent with observations in crossed molecular beam studies and molecular dynamics simulations that suggest that iodine as a leaving group in an SN2 reaction affects the reaction dynamics.
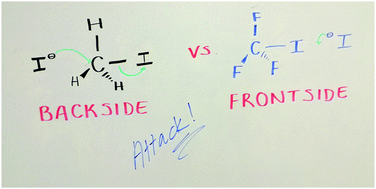
- This article is part of the themed collections: Photodissociation and reaction dynamics and 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...