Self-emitting blue and red EuOX (X = F, Cl, Br, I) materials: band structure, charge transfer energy, and emission energy†
Abstract
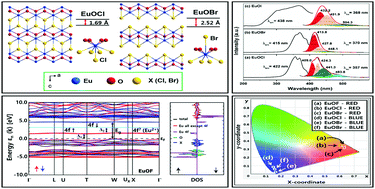
Self-emitting blue and red EuOX (X = F, Cl, Br, and I) were successfully synthesized and characterized. Far-infrared and Raman measurements revealed that the vibration modes prominently reflected the Eu–O and Eu–X bond characters of these materials. X-ray photoelectron spectroscopy (XPS) of the red-emitting EuOX compounds showed that Eu exclusively existed as Eu3+, while in the blue-emitting EuOX, a mixed Eu3+/Eu2+ state was observed. For the red-emitting EuOX (X = F, Cl, and Br), the maximum wavelengths of the charge-transfer (CT) bands were red-shifted: F → Cl → Br (282, 320, and 330 nm for F, Cl, and Br, respectively). Using one-electron spin-polarized band structure calculations, it was verified that the red-shift of the CT energy from F to Br in EuOX was mainly due to the relative positions of the halogen orbital energies being gradually increased, following the trend in their electronegativity. For the blue-emitting EuOX (X = Cl, Br, and I), the emission band maxima were red-shifted from Cl to I (409, 414, and 432 nm for Cl, Br, and I, respectively), which was quite opposite to the trend predicted based on the spectrochemical series in crystal field theory, which was in good agreement with the previous results of the calculated 5d → 4f transition energies of the Eu2+ activator based on the crystal field theory. Through photoluminescence, UV-visible absorbance, and XPS, it was elucidated that the red emission due to Eu3+ was strongly masked by the intensified blue emission associated with the small amount of Eu2+ in the blue-emitting EuOX (X = Cl, Br, and I). These materials may provide a platform for modeling new phosphors for application in solid-state lighting.


 Please wait while we load your content...
Please wait while we load your content...