Mechanisms of photoreactivity in hydrogen-bonded adenine–H2O complexes†
Abstract
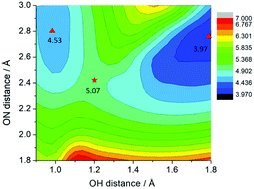
The mechanisms of photoinduced reactions of adenine with water molecules in hydrogen-bonded adenine–water complexes were investigated with ab initio wave-function-based electronic-structure calculations. Two excited-state electron/proton transfer reaction mechanisms have been characterized: H-atom abstraction from water by photoexcited adenine as well as H-atom transfer from photoexcited adenine or the (adenine+H) radical to water. In the water-to-adenine H-atom transfer reaction, an electron from one of the p orbitals of the water molecule fills the hole in the n (π) orbital of the nπ* (ππ*) excited state of adenine, resulting in a charge-separated electronic state. The electronic charge separation is neutralized by the transfer of a proton from the water molecule to adenine, resulting in the (adenine+H)⋯OH biradical in the electronic ground state. In the adenine-to-water H-atom transfer reaction, πσ* states localized at the acidic sites of adenine provide the mechanism for the photoejection of an electron from adenine, which is followed by proton transfer to the hydrogen-bonded water molecule, resulting in the (adenine–H)⋯H3O biradical. The energy profiles of the photoreactions have been computed as relaxed scans with the ADC(2) electronic-structure method. These reactions, which involve the reactivity of adenine with hydrogen-bonded water molecules, compete with the well-established intrinsic excited-state deactivation mechanisms of adenine via ring-puckering or ring-opening conical intersections. By providing additional decay channels, the electron/proton exchange reactions with water can account for the observed significantly shortened excited-state lifetime of adenine in aqueous environments. These findings indicate that adenine possibly was not only a photostabilizer at the beginning of life, but also a primordial photocatalyst for water splitting.
- This article is part of the themed collection: Photodissociation and reaction dynamics


 Please wait while we load your content...
Please wait while we load your content...