Photoluminescence control and abnormal Eu3+ orange emission in Ln3+ (Ln3+ = Ce3+, Eu3+)-doped oxyapatite-type phosphors†
Abstract
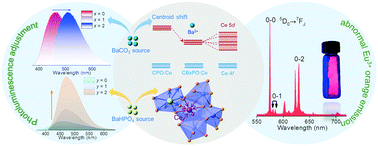
Establishing effective methods to optimize luminescent materials has been a significant challenge for white light-emitting-diode (WLED) applications. Herein, we successfully realized the adjustment of the photoluminescence and enhancement of the thermal stability of Ln3+ (Ln3+ = Ce3+, Eu3+)-doped Ca10−x/yBax/y(PO4)6O phosphors. First, Ba2+ ions from raw BaCO3 material realized a spectral red shift from 464 nm to 506 nm, which we suggested to be due to a synergistic effect from a centroid shift and crystal field splitting. When using BaHPO4 as a raw material, the PL intensity was 6.5 times that of the initial intensity. Surprisingly, the thermal stability was also improved, which was attributed to the increasing lattice distortion introducing many traps. Second, the Ca10(PO4)6O:Eu3+ red phosphor exhibited an abnormal emission phenomenon involving the 5D0 → 7F0 peak at 575 nm becoming the strongest emission peak and the 5D0 → 7F1 peak at 585 nm becoming particularly weak. The site symmetry and charge transfer states were speculated to be closely related to this phenomenon. The charge transfer states were not only correlated with the crystal field strength, but also related to f-electron delocalization, which significantly influenced the energy-level structure of Eu3+. More importantly, according to the relationship between the I(5D0 → 7F0)/I(5D0 → 7F1) ratio and the lattice structure, Eu3+ in the Ca10(PO4)6O compound could be applied as a probe to explore the matrix information, which would provide a helpful method to investigate the lattice structures of compounds and also guide researchers to design new suitable red phosphors.


 Please wait while we load your content...
Please wait while we load your content...