An insight into the non-covalent Pb⋯S and S⋯S interactions in the solid-state structure of a hemidirected lead(ii) complex†
Abstract
A heterodinuclear copper(II)/lead(II) complex with a compartmental ‘reduced Schiff base’ ligand (having inner N2O2 and outer 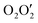 compartments) has been synthesized and characterized. Single-crystal X-ray diffraction analysis confirmed its structure. The X-ray data indicate that the inner N2O2 compartment of the compartmental reduced Schiff base is occupied by copper(II) while the outer
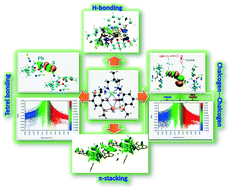
compartments) has been synthesized and characterized. Single-crystal X-ray diffraction analysis confirmed its structure. The X-ray data indicate that the inner N2O2 compartment of the compartmental reduced Schiff base is occupied by copper(II) while the outer 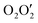 compartment is occupied by lead(II). A thiocyanate anion is coordinated to the copper(II) center via its N-end, whereas, another thiocyanate ion is linked to the lead(II) center via its S-end and is semi-coordinated to the copper(II) center through its N-end. A hemidirectionally coordinated lead(II) center is well suited for establishing tetrel bonding interactions. We estimated the BSSE (basis set superposition error) corrected energies of non-covalent S⋯S, Pb⋯π, and π⋯π interactions and N–H hydrogen bonding along with tetrel bonding by DFT calculations. To obtain an insight into the physical nature of these bonds, we extensively used Bader's quantum theory of atoms-in-molecules (QTAIM). Additionally, the non-covalent interaction reduced density gradient (NCI-RDG) method established nicely the presence of such non-covalent intermolecular interactions. Here, we also used natural bond orbital (NBO) analysis to find out the origin of S⋯S and Pb⋯S bonding.
compartment is occupied by lead(II). A thiocyanate anion is coordinated to the copper(II) center via its N-end, whereas, another thiocyanate ion is linked to the lead(II) center via its S-end and is semi-coordinated to the copper(II) center through its N-end. A hemidirectionally coordinated lead(II) center is well suited for establishing tetrel bonding interactions. We estimated the BSSE (basis set superposition error) corrected energies of non-covalent S⋯S, Pb⋯π, and π⋯π interactions and N–H hydrogen bonding along with tetrel bonding by DFT calculations. To obtain an insight into the physical nature of these bonds, we extensively used Bader's quantum theory of atoms-in-molecules (QTAIM). Additionally, the non-covalent interaction reduced density gradient (NCI-RDG) method established nicely the presence of such non-covalent intermolecular interactions. Here, we also used natural bond orbital (NBO) analysis to find out the origin of S⋯S and Pb⋯S bonding.


 Please wait while we load your content...
Please wait while we load your content...