The multiple roles of carbonic anhydrase in calcium carbonate mineralization†
Abstract
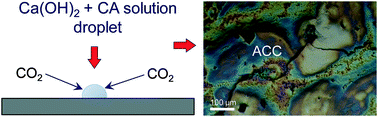
Carbonic anhydrase (CA), a ubiquitous enzyme that catalyzes the reversible hydration of CO2, is known to be involved in the formation of CaCO3 biominerals and is currently used for biomimetic CO2 mineral sequestration. However, its specific role(s) in calcium carbonate (bio)mineralization is poorly understood. Here we show that CA catalyzes the formation of the reactive precursors (i.e., HCO3− and CO32− ions) required for mineralization, accelerates the precipitation of metastable amorphous calcium carbonates and their solution-mediated conversion into crystalline calcite, which grows via a non-classical nanoparticle aggregation mechanism that facilitates CA occlusion. Ca+ and CO32− ions promote the partial unfolding and oligomerization of CA, resulting in fibril- and sheet-like supramolecular assemblies that template nanostructured calcium carbonate crystallization. By losing its catalytic activity following the observed conformational changes, CA elicits a mechanism for arresting calcium carbonate mineralization. Our results show that CA can play multiple, until now unrecognized, critical roles, namely, as an enzyme and structural protein in CaCO3 biomineralization and also helps to explain the observed loss of enzymatic activity during ex situ CO2 mineral sequestration.


 Please wait while we load your content...
Please wait while we load your content...