Facile fabrication of cobalt-doped SnO2 for gaseous ethanol detection and the catalytic mechanism of cobalt†
Abstract
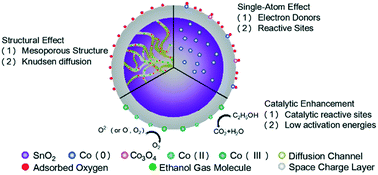
Semiconductors are widely used as basic materials in commercial ethanol detection sensors. In this work, SnO2 sensors were modified by a Co doping method to obtain Co-SnO2 for gaseous ethanol detection. For investigating the best sensing performance and elucidating the underlying catalytic mechanism, the effects of the Co/Sn feed ratio and calcination temperature during sample preparation were examined. The sample with a Co/Sn feed molar ratio of 1.0 calcined at 500 °C (D3-Co-SnO2) possessed better ethanol sensitivity than other samples. The best response to 100 ppm gaseous ethanol (i.e., ratio of sensor resistance in air to that in ethanol) was 105.2 at an operating temperature of 350 °C. More importantly, D3-Co-SnO2 achieved a low detection limit of 1 ppm and apparently higher selectivity over various gases (e.g., acetone, hydrogen, methane, etc.). The enhanced sensing performance was attributed to (1) the incorporation of Co atoms, which increased the number of oxygen vacancies and active sites; (2) the catalytic activity of Co(III) and Co(II), which changed the reaction process; and (3) the suitable pore size, which led to effective diffusion of the gas molecules.


 Please wait while we load your content...
Please wait while we load your content...