Possible pre-phase transition of the α-HMX crystal observed by the variation of hydrogen-bonding network under high pressures†
Abstract
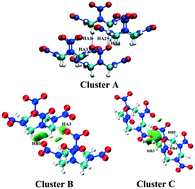
DFT calculations were performed to investigate the pressure-induced hydrogen bond network in the α-HMX crystal in terms of geometrical evolution, electronic structure, Hirshfeld surface, AIM method, IGM , mutual penetration distance, mechanical properties, and vibrational spectra. The results show that the hydrogen bond network predominates in the intermolecular interactions in the crystal and becomes more sensitive in the low pressure region (below 10 GPa). Compared to the relatively strong C–H⋯O interactions related to vertical nitro groups, the weak hydrogen bonds located on horizontal N–NO2 are more complicated and may govern the physicochemical properties of the metastable α-HMX crystal under compression. The abrupt variation of the hydrogen bond network in the crystal at 4 GPa that is attributed to shifting of relative position of the molecules indicates the emergence of possible pre-phase transition before the transformation from β- to δ-HMX. Our investigations uncover detailed variation of noncovalent interactions in the α-HMX crystal at high pressures, which provides a deeper understanding of the safety performance of nitramine explosives.


 Please wait while we load your content...
Please wait while we load your content...