Influence of counterions on the supramolecular frameworks of isoquinoline-based silver(i) complexes†
Abstract
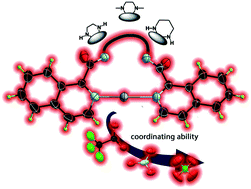
Silver coordination compounds have been widely studied, among others, because of their fascinating structures where the counterions can play an essential role in the construction of supramolecular networks. Herein, seven new Ag(I) complexes containing bis(1-isoquinolinecarboxamide)ethane (L1), bis(1-isoquinoline-N,N′-dimethylcarboxamide)ethane (L2) and bis(1-isoquinolinecarboxamide)propane (L3) as ligands have been synthesized and their crystal structures were obtained by single crystal X-ray diffraction, with ClO4−, NO3− and CF3CO2− as counterions. These species were analysed from three points of view: first, the differences and similarities among the amide-to-amide hydrogen bond patterns when compared to those of the bis(1-isoquinolinecarboxamide)alkane ligands; second, their structural patterns based on the coordinative ability of the counterions; finally, the analysis of the robustness of the amide-to amide intermolecular hydrogen bonds when a coordination complex is formed. Among the most critical features of anions, it is their coordinating ability and participation in non-covalent interactions with the ligands that are contextualized in this research.


 Please wait while we load your content...
Please wait while we load your content...