Visible-light CO2 photoreduction of polyoxometalate-based hybrids with different cobalt clusters†
Abstract
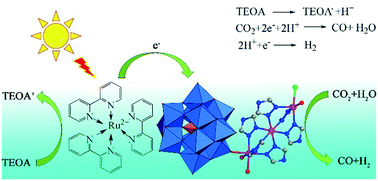
The photoreduction of carbon dioxide (CO2) into a valuable energy gas (CO and H2) is an efficient approach to address the fossil fuel crisis and mitigate the global warming effect. The development of effective photocatalysts for CO2 reduction is still desirable and challenging. Herein, two novel polyoxometalate-based hybrids with multinuclear cobalt clusters, [Co2.67(SiW12O40)(H2O)4(Htrz)4]·Cl1.33 (Htrz = 1,2,4-triazole) (1) with a binuclear cobalt cluster and [Co3(SiW12O40)(H2O)3(Htrz)6Cl]·Cl·6H2O (2) with a trinuclear cobalt cluster, were synthesized under hydrothermal conditions. Both of them were characterized by single-crystal X-ray diffractions, PXRD, IR, TG and UV–vis spectra. Compound 1 exhibited a 3D structure with 4-connected [SiW12O40] and 3-connected [Co2(Htrz)3(H2O)3] secondary building units (SBUs). The 1D chain of compound 2 was constructed from [SiW12O40] and [Co3(Htrz)6(H2O)3Cl] SBUs. Furthermore, the photoreduction of CO2 under visible light by the two cobalt-based POMs was investigated using [Ru(bpy)3]Cl2·6H2O as a photosensitizer. The CO yields of compounds 1 and 2 were 15 705 and 18 501 μmol g−1 for the CO2 photocatalytic reduction under three hour irradiation at 293 K, respectively. The difference in the photocatalytic performance of 1 and 2 was explained by comparing the energy of the valence band, band gaps and conduction band. The results showed that the photocatalysts incorporated with multinuclear Co clusters could effectively improve photocatalytic activities, thus providing a valuable view to design high performance and cost-acceptable molecular catalysts for CO2 photoreduction.


 Please wait while we load your content...
Please wait while we load your content...