Facile hydrothermal synthesis of hierarchical porous priceite (Ca4B10O19·7H2O) microspheres as high-efficiency adsorbents for heavy metal ions removal†
Abstract
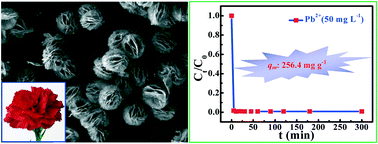
Heavy metal ions have attracted considerable concern for their high toxicity and potential malign effects on the water environment, and adsorption has been regarded as one of the most versatile and effective techniques towards water treatment. In view of this, hierarchical porous carnation-like Ca4B10O19·7H2O microspheres self-assembled by two-dimensional (2D) quasi-hexagonal nanoplates are successfully synthesized for the first time as far as we know, by a facile and surfactant ethylenediaminetetraacetic acid disodium salt (EDTA-2Na) assisted hydrothermal approach using CaCl2·2H2O, NaOH and H3BO3 as the raw materials. The as-obtained carnation-like Ca4B10O19·7H2O microspheres exhibit a uniform morphology, ca. 89% of which have a diameter within the range of 8.0–11.0 μm. A probable EDTA-2Na assisted self-assembly formation mechanism has also been proposed based on systematic control experiments. Meanwhile, the carnation-like Ca4B10O19·7H2O microspheres demonstrate a transparent characteristic from the ultraviolet to the visible region, a hierarchical porous structure (specific surface area: 32.79 m2 g−1, pore volume: 0.151 cm3 g−1, average pore diameter: 3.704 nm) and satisfactory thermal stability. In addition, the carnation-like microspheres are confirmed as great satisfactory adsorbents for heavy metal ion (Pb2+) removal with an adsorption capacity qm of 256.4 mg g−1, which may also be employed as excellent adsorbent candidates for other heavy metal or organic dye removal in water treatment as well as catalyst carriers in heterogeneous catalysis.


 Please wait while we load your content...
Please wait while we load your content...