Hydrothermal synthesis of natroalunite nanostructures and their F−-ion removal properties in water†
Abstract
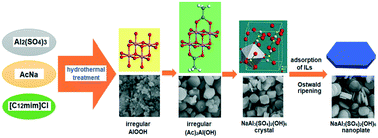
Natroalunite nanostructures with various morphologies have been synthesized by an ionic liquid-assisted hydrothermal route at relatively low temperatures and their F− ion adsorption characteristics have been studied. The possible formation and growth mechanism of alunite crystallites by the ionic liquid-assisted hydrothermal process was proposed and discussed in detail. The reaction conditions, such as the presence and the amount of templating agent (ionic liquid), the reaction temperature and the composition of the reaction system, play considerable roles on the structure and morphologies of the final products. Our results lead to the assumption of rapid nucleation of AlOOH and subsequent ionic liquid-templated growth of NaAl3(SO4)2(OH)6 nanostructures. In these processes, irregular AlOOH particles are firstly formed, and the final natroalunite crystals are obtained through crystal phase conversions from AlOOH to (CH3COO)2AlOH (Ac2AlOOH) and then to NaAl3(SO4)2(OH)6. With the increasing amount of ionic liquid and reaction temperature, the morphology of the products was changed from irregular to well-developed nanostructures, such as cylindrical, round sheet-like, flower-like, microspherical and hexagonal nanoplates. The transformation of Ac2AlOOH into natroalunite NaAl3(SO4)2(OH)6 is a reversible reaction. Therefore, as the amount of AcNa increases, the amount of intermediates included in the final product increases. The BET specific surface area of the natroalunite product was found to be as high as about 364.0 m2 g−1 and the maximum fluoride adsorption capacity of it was 88.58 mg g−1 at pH 7.0.


 Please wait while we load your content...
Please wait while we load your content...