Chloranilate bridged dinuclear copper(ii) complexes: syn–anti geometry tuned by the steric factor and supramolecular interactions†
Abstract
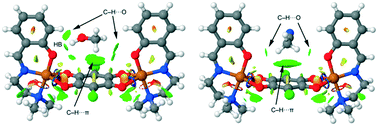
Four dinuclear Cu(II) coordination compounds, [(CuL1)2CA]·CH3OH (1), [(CuL1)2CA]·CH3CN (1A), [(CuL2)2CA] (2) and [(CuL3)2CA] (3) (where, CA = chloranilate anion, HL1 = 2-[1-(2-dimethylamino-ethylimino)-methyl]-phenol, HL2 = 2-[1-(2-methylamino-ethylimino)-methyl]-phenol, HL3 = 2-[1-(methylamino-propan-2-ylimino)-ethyl] phenol, have been synthesized by the self-assembly of copper–Schiff base precursors together with chloranilate anions as bridging ligands. The complexes are characterized by elemental analyses, IR spectroscopy, thermal analyses, and single crystal X-ray crystallography. Structural analyses reveal that complexes 1 and 1A are in the syn-conformation while 2 and 3 are in the anti-conformation with respect to the metal coordinated Schiff-base ligands. Theoretical calculations show that for all three ligands, the anti-conformations are energetically favorable and the unfavourable syn-conformation in 1 or 1A is stabilized through CH⋯π and hydrogen bonding interactions between the [(CuL1)2CA] unit and a co-crystallised solvent molecule. The M06-2X/def2-TZVP level of theory and NCI plot explicitly show that unlike in the case of 2 and 3, which form a 1D supramolecular chain by chelate ring⋯chelate ring π-stacking and cuprophilic interactions between neighbouring dimers, in 1 and 1A, steric interactions between neighbouring dimeric units, incorporated by one extra methyl group of the Schiff-base moiety, restrict these structures into dimeric units. Variable-temperature magnetic susceptibility measurements suggest weak antiferro- and ferromagnetic coupling in 1 (syn-conformation) and 3 (anti-conformation) with coupling constants of −2.11 and 3 cm−1, respectively.


 Please wait while we load your content...
Please wait while we load your content...