Three coordination polymers based on 3-(3′,5′-dicarboxylphenoxy)phthalic acid and auxiliary N-donor ligands: syntheses, structures, and highly selective sensing for nitro explosives and Fe3+ ions†
Abstract
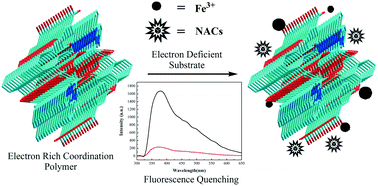
Three new coordination polymers derived from a 3-(3′,5′-dicarboxylphenoxy)phthalic acid (H4L) ligand have been constructed with Zn(II) and Cd(II) ions under hydrothermal conditions, namely [Zn(H2L)(2,2-bipy)]n (1), [Cd(H2L)(phen)(H2O)]n (2), and [Cd(H2L)(4,4-bipy)]n (3) (2,2′-bipy = 2,2′-bipyridine, phen = 1,10-phenanthroline hydrate, 4,4′-bipy = 4,4′-bipyridine). All the three compounds were characterized by single-crystal X-ray diffraction, elemental analysis, FT-IR, powder X-ray diffraction and thermogravimetric analyses. Compounds 1 and 2 are respectively driven by hydrogen bonding and π–π stacking interactions to form 3D supramolecular networks. Compound 3 exhibits a 2D layer network, and then shows a 2D → 3D structure through C–H⋯O hydrogen bonds. The photoluminescence properties of compounds 1–3 in the solid state are also investigated. Detailed property investigation shows that compound 1 can serve as a promising platform for luminescence sensing, as it has high selectivity and sensitivity toward Fe3+ anions in the solution and high detection for the trace content of 2,4-dinitrophenol with a high Ksv value of 1.83 × 104 M−1 and a low detection limit of 7.08 × 10−4 mM.


 Please wait while we load your content...
Please wait while we load your content...