Eco-friendly nanostructured Zn–Al layered double hydroxide photocatalysts with enhanced photocatalytic activity†
Abstract
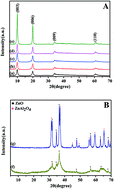
Zn–Al layered double hydroxides (Zn–Al LDHs) with various reaction times were synthesized by a hydrothermal method at constant pH, and they were tested for the removal of RhB with or without post-calcination treatment. The effect of reaction time and calcination treatment on the structures, morphologies, chemical compositions and photocatalytic properties of Zn–Al LDHs was analyzed by XRD, TG/DSC, SEM, XPS, FTIR, Raman, PL and UV-vis DRS in detail. The optimized Zn–Al LDHs with a book page morphology had good crystal quality and the sample morphology changed to irregular flakelets with increasing calcination temperature. The RhB photodegradation results showed that the photocatalytic activity of Zn–Al LDHs was enhanced with increasing reaction time due to the improved crystal quality and the better separation of photogenerated electron–hole pairs. It was found that the calcination temperature was also an important factor for the c-Zn–Al-LDH samples, and the c-Zn–Al-LDH-24-300 sample exhibited the best photocatalytic activity due to the highest efficiency of photogenerated electron–hole separation, especially the formation of a new phase. A pseudo first order rate and recycling experiments were also used to analyze the photocatalytic activity.


 Please wait while we load your content...
Please wait while we load your content...