Revealing the role of a surfactant in the nucleation and crystal growth of thiamine nitrate: experiments and simulation studies
Abstract
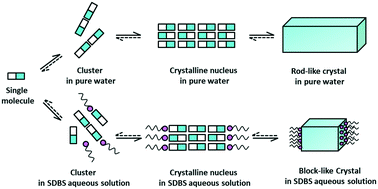
In this work, the influence of sodium dodecyl benzene sulfonate (SDBS) on the primary nucleation and growth of thiamine nitrate (VB1) has been investigated. At a given supersaturation, SDBS significantly impedes the nucleation rate of VB1. The results were analyzed through the probability distribution function of the induction time. Furthermore, the growth rate of VB1 in pure water and in the presence of SDBS was determined by single seed crystal growth experiments. It was found that the retardation by SDBS of the axial direction growth of VB1 crystals is much stronger than that of the radial direction growth. Molecular dynamics simulations (MD) were employed to delineate the effects of SDBS on the nucleation and growth of VB1. The MD simulation results manifested that the strong steric hindrance of SDBS molecules, the hydrogen-bonding and electrostatic interactions between SDBS molecules and VB1 molecules hinder the nucleation rate of VB1. Moreover, the results revealed the highest bonding energy between SDBS and the (1 0 0) surface, and the bonding energies between SDBS and all corresponding surfaces obey the following trend: (1 0 0) > (1 1 0) > (1 1 1) > (1 0 2) > (1 0 −2) > (0 1 1) > (0 0 2), which can provide a reasonable explanation for the experimental results.


 Please wait while we load your content...
Please wait while we load your content...