Conformational variation of ligands in mercury halide complexes; high and low Z′ structures†
Abstract
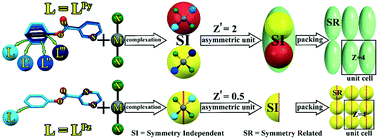
In this study, five new mercury halide complexes of [Hg(LPy)2Cl2] (1), [Hg(LPy)2Br2] (2), [Hg(LPy)2I2] (3), [Hg(LPz)2Cl2] (4) and [Hg(LPz)2Br2] (5) in which LPy is a phenyl pyridine-2-carboxylate and LPz is a phenyl pyrazine-2-carboxylate, were designed and synthesized. The complexes were characterized using thermal gravimetric analysis (TGA), differential scanning calorimetry (DSC), CHN-elemental analyses, Fourier transform infrared spectroscopy (FT-IR), NMR spectroscopy, powder X-ray diffraction (PXRD), and single crystal X-ray diffraction (SCXRD). The asymmetric unit of compounds 1–3 contains two independent complexes (high Z′) and compounds 4 and 5 crystallized with half of the complex in the asymmetric unit (low Z′). Interestingly, structural analysis revealed that small changes in the ligand molecules (Py to Pz) affect the number of molecules in the asymmetric unit. Indeed, conformational variation of the ligand (LPy), which is related to the rotation of the phenyl ring, forms four symmetry-independent (SI) ligands of the complex structures in 1–3. In contrast, there is no conformational change for Lpz in complexes 4 and 5. The C–H⋯O, C–H⋯X–M, and C–H⋯π interactions have a major role in the aggregation of molecular structures in the titled compounds. The roles of the intermolecular interactions in the conformational variation of ligands were investigated using theoretical studies and Hirshfeld surface analysis on the SI and symmetry-related (SR) molecular pairs. The results show that C–H⋯O/X–M have a key role in the stabilization of the SI motifs in 1–3. The conformational variation in the complexes were examined based on the rmsd of the overlay for molecules in the asymmetric unit for inorganic high and low Z′ pairs which are presented in the Cambridge Structural Database (CSD). The flexibility of ligands in the complexes is introduced as an effective factor for structural variation of molecules in the asymmetric unit. In this regard, the high Z′ complexes of 1–3 belong to the essential conformational adjustment class of SI molecules. In fact, phenyl ring rotation, which was previously been reported as a reason for conformational polymorphism in the LPy free ligand, is suggested as a reasonable element for high Z′ in 1–3. Conversely, pyrazine rotation in the LPz free ligand, which was previously reported as being responsible for making different polymorphs, did not occur in complexes 4 and 5 owing to coordination of this moiety to a mercury metal centre and later appeared in the low Z′ form of the structures.


 Please wait while we load your content...
Please wait while we load your content...