Commenting on the photoreactions of anthracene derivatives in the solid state
Abstract
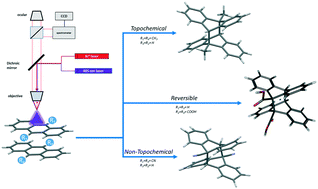
We review a series of photochemical reactions related to the renowned family of anthracene derivatives. We first classify the photodimerizations of 9-methyl-anthracene, 9-cyano-anthracene and 9-anthracene-carboxylic acid as topochemical, non-topochemical and reversible, respectively. We then introduce a chemical reaction where the lattice change involves two different molecular species. In the latter case, the compatibility of the reactive species occurs at the level of symmetry matching of the unit cells rather than via a correct registry of molecular orientations. We then describe the relationship between incoming photons and mechanical motion, a common occurrence in crystal-to-crystal photoreactions, which shows stunning effects when 9,10-dinitro-anthracene is transformed to anthraquinone. Finally, the kinetics of these systems is shortly discussed. By means of a micro-Raman technique, we are capable of monitoring, at the same time and on the very same spot, the molecular (intramolecular vibrations or internal motions) and the lattice (intermolecular vibrations or lattice phonons) transformations. It turns out that molecular changes start first, producing, in a following time, the symmetry modifications of the crystal lattice. The extent of the delay of the structural change may considerably vary depending upon the system studied.
- This article is part of the themed collection: 2019 Highlight article collection


 Please wait while we load your content...
Please wait while we load your content...