3D structured Mn2O3 synthesized using tween surfactant: influence on the morphology and oxygen reduction catalytic performance
Abstract
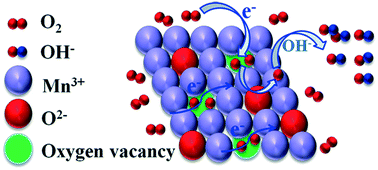
3D structured Mn2O3 with flower-like (Mn2O3-F), globe-like (Mn2O3-G) or rhombohedral (Mn2O3-R) morphologies were successfully synthesized by solvothermal method by controlling the amounts of Tween surfactant. These Mn2O3 oxides enhance the catalytic activity for the oxygen reduction reaction (ORR) in direct methanol fuel cells (DMFCs) significantly, while PtRu/C works as the anode catalyst and polymer fiber membrane (PFM) functions as the electrolyte film. Mn2O3-G shows higher ORR catalytic activity than Mn2O3-F and Mn2O3-R. The maximum power density (Pmax) of Mn2O3-F-, Mn2O3-G- and Mn2O3-R-based DMFCs are 11.44, 21.03 and 10.22 mW cm−2 at room temperature, respectively. The influence of the Tween surfactant on the microstructure and ORR properties were also studied systematically. Compared with Mn2O3-F and Mn2O3-R, the Raman and XPS results indicate that Mn2O3-G has more oxygen vacancies, which can promote the electrical conductivity and act as an active center to absorb and store available O2 for stable ORR properties.


 Please wait while we load your content...
Please wait while we load your content...