Calcium stannyl formation by organostannane dehydrogenation†
Abstract
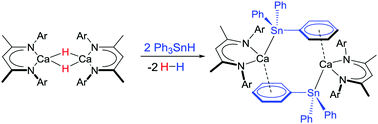
Reaction of the dimeric calcium hydride, [(BDI)CaH]2 (1), with Ph3SnH ensues with elimination of H2 to provide [(BDI)Ca-μ2-H-(SnPh3)Ca(BDI)] (3) and [(BDI)Ca(SnPh3)]2 (4) alongside dismutation to Ph4Sn, H2 and Sn(0). DFT analysis indicates that stannyl anion formation occurs through deprotonation of Ph3SnH and with retention of dinuclear species throughout the reactions.


 Please wait while we load your content...
Please wait while we load your content...