Divergent total syntheses of five illudalane sesquiterpenes and assignment of the absolute configuration†
Abstract
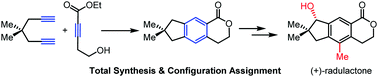
Concise, divergent total syntheses of five bioactive illudalane sesquiterpenes have been achieved. Our synthesis features an intermolecular [2+2+2] cycloaddition, and a lactone-directed aromatic C–H oxygenation to generate a temporary phenolic hydroxyl group which enables regioselective methylation. Furthermore, the absolute configuration of radulactone was assigned by chemical synthesis.
- This article is part of the themed collections: Celebrating a Century of Excellency in Chemistry at Xiamen University and Natural product synthesis


 Please wait while we load your content...
Please wait while we load your content...