Topochemical nitridation of Sr2FeMoO6†
Abstract
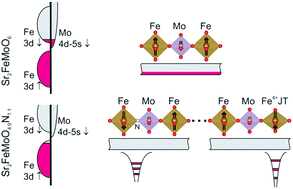
The topotactic nitridation of cation ordered, tetragonal Sr2FeMoO6 in NH3 at moderate temperatures leads to cubic, Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m double perovskite oxynitride Sr2FeMoO4.9N1.1 where double-exchange interactions determine ferromagnetic order with TC ≈ 100 K. Substitution of oxide by nitride induces bond asymmetries and local electronically driven structural distortions, which combined with Fermi level lowering restricts charge itinerancy to confined regions and preclude spontaneous long-range magnetic order. Under a magnetic field, ferromagnetic correlations expand, favoring charge delocalization and a negative magnetoresistance is observed.
m double perovskite oxynitride Sr2FeMoO4.9N1.1 where double-exchange interactions determine ferromagnetic order with TC ≈ 100 K. Substitution of oxide by nitride induces bond asymmetries and local electronically driven structural distortions, which combined with Fermi level lowering restricts charge itinerancy to confined regions and preclude spontaneous long-range magnetic order. Under a magnetic field, ferromagnetic correlations expand, favoring charge delocalization and a negative magnetoresistance is observed.
- This article is part of the themed collection: Perovskites


 Please wait while we load your content...
Please wait while we load your content...