The role of oxygen vacancies in water oxidation for perovskite cobalt oxide electrocatalysts: are more better?†
Abstract
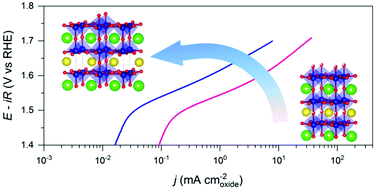
We explore the role of oxygen vacancies in the oxygen evolution reaction (OER) for double perovskite PrBaCo2O6−δ. Interestingly, we find that largely increasing oxygen vacancies leads to a significant reduction in the intrinsic OER activity. Structural studies reveal that oxygen vacancies tend to orderly align in PrO1−δ. This ordered structure not only lowers the cobalt oxidation states but also triggers a spin-state transition from high-spin to low-spin states for cobalt ions, both greatly slowing the OER kinetics.


 Please wait while we load your content...
Please wait while we load your content...