Collaborative assembly of doxorubicin and galactosyl diblock glycopolymers for targeted drug delivery of hepatocellular carcinoma†
Abstract
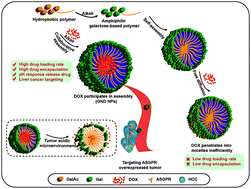
Hepatocellular carcinoma (HCC) patients suffer from severe pain due to the serious systemic side effects and low efficiency of chemotherapeutic drugs, and it is important to develop novel drug delivery systems to circumvent these issues. In this study, a series of galactose-based glycopolymers, poly(N-(prop-2-enoyl)-β-D-galactopyranosylamine)-b-poly(N-isopropyl acrylamide) (pGal(OH)-b-pNIPAA), were prepared through a sequential reversible addition-fragmentation chain transfer (RAFT) polymerization and tetrabutylammonium hydroxide (TBAOH)-mediated removal of acetyl groups. Hydrophilic doxorubicin hydrochloride was introduced to undergo collaborative assembly with poly(N-(prop-2-enoyl)-β-D-peracetylated galactosamine)-b-poly(N-isopropyl acrylamide) (pGal(Ac)-b-pNIPAA) via TBAOH treatment. pGal-b-pNIPAA/doxorubicin (DOX) delivery nanoparticles (GND NPs) formed by collaborative assembly were fully characterized by NMR, TEM and FT-IR, indicating the well-controlled formation of particles with uniform size and high efficiency in terms of drug loading and encapsulation compared with conventional adsorption methods. Meanwhile, the GND NPs were observed to be rapidly disintegrated under acidic conditions and resulted in an increased release of DOX. Cellular experiments showed that pGal-b-pNIPAA/DOX is apparently an asialoglycoprotein receptor (ASGPR)-mediated target of HCC, resulting in enhanced cellular uptake to HepG2 cells and anti-tumor efficacy in vitro. Furthermore, GND NPs III exerted more sustainable and effective anti-tumor effects compared to free DOX on a transgenic zebrafish TO(KrasG12V) model in vivo. These results indicated that the biocompatible nanomaterials developed by collaborative assembly with galactosyl diblock glycopolymers and DOX may serve as a promising candidates for targeting therapy of HCC.


 Please wait while we load your content...
Please wait while we load your content...