Microenvironment-activated nanoparticles for oxygen self-supplemented photodynamic cancer therapy†
Abstract
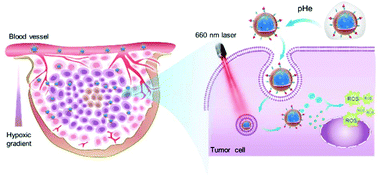
Tumor hypoxia, as a hallmark of most solid tumors, poses a serious impediment to O2-dependent anticancer therapies, such as photodynamic therapy (PDT). Although utilizing nanocarriers to load and transport O2 to tumor tissues has been proved effective, the therapeutic outcomes have been impeded by the low O2 capacity and limited tumor penetration of the nanocarriers. To address these problems, we incorporated perfluorooctyl moieties into nanocarriers to improve the encapsulation of perfluorooctyl bromide via fluorophilic interactions, leading to elevated O2 capacity of the nanocarriers. Meanwhile, to enhance the tumor cell penetrating ability as well as reduce reticuloendothelial system recognition, the nanocarrier was further decorated with a cell-penetrating peptide, which was masked with a protecting group via an acid-labile amide bond for prolonged circulation time and acid-activated cell penetration. The in vitro study demonstrated that, apart from remarkably boosting the photocytoxicity of chlorin 6 (Ce6) at a low dosage, the rationally designed O2@DANPCe6+PFOB could even alleviate the pre-existing tumor hypoxia. After intravenous injection, O2@DANPCe6+PFOB exhibited significant tumor accumulation and retention, and potent tumor growth inhibition compared to traditional PDT. Overall, the O2@DANPCe6+PFOB mediated O2 self-supplemented PDT with tumor acidic microenviornment-activated cell penetration provides a promising strategy in anticancer treatment.


 Please wait while we load your content...
Please wait while we load your content...