Multifunctional theranostic nanosystems enabling photothermal-chemo combination therapy of triple-stimuli-responsive drug release with magnetic resonance imaging†
Abstract
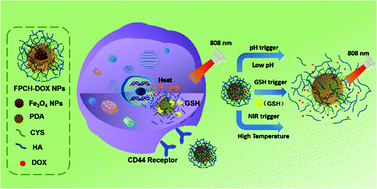
Theranostic nanosystems are emerging as a promising approach for controlled drug delivery, diagnosis and multimodal therapeutics. Herein, a multifunctional theranostic nanoplatform is reported for photothermal-chemo combination therapy functioned with magnetic and thermal imaging. Hyaluronic acid (HA) coated Fe3O4@polydopamine nanoparticles equipped with redox-sensitive disulfide linkers have been subsequently deposited with an anticancer drug, doxorubicin (DOX) (termed as FPCH-DOX NPs). These nanocomposites possess an average diameter of 120 nm, a saturation magnetization of 28.5 emu g−1, DOX loading capacity of 7.13% and a transverse relaxation rate of 171.76 mM−1 s−1. The drug release could be triggered by pH, glutathione (GSH) concentration and light irradiation. Prussian blue staining and confocal microscopy demonstrate that these nanoplatforms have improved biocompatibility and cellular uptake in CD44-positive HeLa cell lines rather than in CD44-negative NIH 3T3 normal cell lines. In vitro evaluations demonstrate that the combination therapy of FPCH-DOX NPs lowers the cell viability to 16.2%, less than that of individual chemotherapy (55.3%) or PTT (52.1%). In vivo MRI indicates that the tumor accumulation of FPCH-DOX NPs provides enhanced MRI contrast, and in vivo thermal imaging verified their localized photothermal conversion effect in tumor tissues. Importantly, FPCH-DOX NPs present remarkable anti-tumor efficacy by photothermal-chemo combination therapy. H&E and Ki67 staining tests show obvious necrosis and weak cell proliferation at the region of the tumor. Thus, FPCH-DOX NPs are promising multifunctional nanoplatforms for highly effective cancer theranostics.


 Please wait while we load your content...
Please wait while we load your content...