Sustained protein therapeutics enabled by self-healing nanocomposite hydrogels for non-invasive bone regeneration†
Abstract
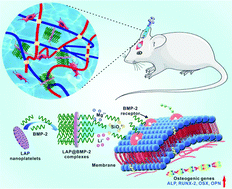
Bone tissue engineering based on stem cells, growth factors and bioactive scaffolds presents an appealing but challenging approach for rehabilitation of patients with bone defects. A versatile system with the capability for easy operation and precise protein delivery in specific locations is attractive for enhancing bone regeneration. Here, we develop a non-invasive delivery system based on injectable and self-healing nanocomposite hydrogels for sustained protein release, which has the potential to improve the current orthopedic strategy. Specifically, LAPONITE® (LAP) nanoplatelets are able to accelerate the gelation process through hydrogen bonds with polysaccharide matrices, endowing hydrogels with superior mechanical and rheological behaviors, along with better injectability and self-healing ability. Attractively, the strong static binding between LAP nanoplatelets and bone morphogenetic protein-2 (BMP-2) can form stable LAP@BMP-2 complexes. The results indicate that the complexes effectively preserve the intrinsic bioactivity of BMP-2 and prolong the release period for more than four weeks. Moreover, hydrogels incorporating with the LAP@BMP-2 complexes synergistically boost cell spreading, proliferation activity and osteogenesis, both in vitro and in vivo, compared with LAP or BMP-2 alone. Overall, this study proposes a valid platform for protein therapeutics and non-invasive bone repair.


 Please wait while we load your content...
Please wait while we load your content...