Coadministration of chemotherapy and PI3K/Akt pathway treatment with multistage acidity/CathB enzyme-responsive nanocarriers for inhibiting the metastasis of breast cancer†
Abstract
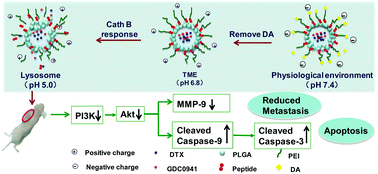
As the principal reason for the inducement of high mortality, tumor metastasis is regulated by different pathways owing to its complexity and multistep process. In order to inhibit the proliferation and metastasis of human breast cancer simultaneously, controlling the codelivery of chemotherapeutics and pathway inhibitors precisely has been considered as a high-potential strategy to accurately eliminate tumor metastasis. In this study, polymer PLGA-p-PEI-DA was synthesised and automatically assembled into a cascade “trinity” response drug delivery system, i.e., PPP-DA/NPs (PLGA: poly(lactin-co-glycolic acid), PEI: polyethyleneimine, DA: 2,3-dimethylmaleic anhydride). In the tumor microenvironment, PPP-DA/NPs could remove the outer DA molecules via the pH-sensitive hydrolysis of β-carboxylic amide bonded with DA and PEI. Then, PPP-DA/NPs were broken up owing to the enzymatically cleavable GFLGF (Gly-Phe-Leu-Gly-Phe) linker. The structure of the polymer and the properties of PPP-DA/NPs were evaluated in detail. Moreover, studies on the antitumor metastasis efficiency and antitumor mechanism of PPP-DA/NPs were carried out in detail. As demonstrated in this study, PPP-DA/NPs could reverse the potential in pH 6.8 PBS and showed elevated cellular uptake efficiency. Moreover, PPP-DA/NPs exhibited strong antitumor metastasis ability in vitro and in vivo. The tumor inhibiting rate (TIR) of PPP-DA/NPs (68.4%) was significantly higher than that of docetaxel (DTX) (5.9%). The antitumor mechanistic studies confirmed that PPP-DA/NPs could down-regulate the expressions of Akt, MMP-9 and pro-caspase-3/9 protein, as indicated by western blot analysis. This multifunctional drug delivery system (DDS) is highly selective and effective in inhibiting tumor metastasis, which shows a great potential in inventing smart nanocarriers for targeted tumor-metastasis therapy.

 Please wait while we load your content...
Please wait while we load your content...