Ultralong circulating choline phosphate liposomal nanomedicines for cascaded chemo-radiotherapy†
Abstract
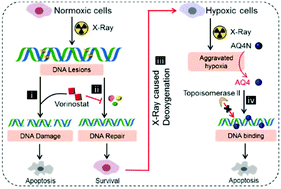
Cancer radiation therapy (RT) is limited by endogenous DNA repair of tumor cells and microenvironmental hypoxia in tumor tissues. Herein, we demonstrated an effective cancer chemo-radiotherapy strategy based on choline phosphate liposomal nanomedicines, which inhibit the intrinsic radioresistance of RT and concomitantly harness the RT-induced hypoxia to produce additional toxicity to overcome post-RT radioresistance. To achieve this strategy, a radiotherapy sensitizer, vorinostat, and a hypoxia-activated banoxantrone dihydrochloride (AQ4N) were simultaneously delivered to a tumor using liposomes composed of an inverted polarity lipid 2-((2,3-bis(oleoyloxy)propyl)dimethylammonio)ethyl ethyl phosphate (DOCPe). The DOCPe liposomes exhibited a longer blood circulation time and enhanced tumor accumulation, compared to their zwitterionic phosphocholine counterpart. The RT was sensitized by vorinostat to kill non-tolerant normoxic tumor cells efficiently. The irradiation aggravated hypoxia-activated AQ4N to further potentiate RT treatment. This chemo-radiotherapy combination showed excellent tumor treatment efficacy and is promising for future clinical translation.


 Please wait while we load your content...
Please wait while we load your content...