Quantitative LC-MS/MS method for nivolumab in human serum using IgG purification and immobilized tryptic digestion†
Abstract
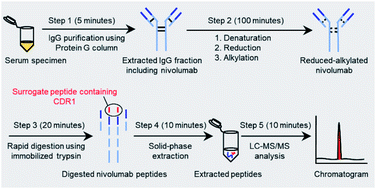
A liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS) method is a novel approach for the quantification of therapeutic monoclonal antibody. However, this method has severe ionization suppression and time-consuming sample preparation. In the present study, we developed a simple and rapid LC-MS/MS method for quantifying serum nivolumab with good analytical performance using immunoglobulin G (IgG) purification and immobilized tryptic digestion. Surrogate peptide derived from nivolumab was identified by a hybrid quadrupole-Orbitrap mass spectrometer. IgG purification and immobilized tryptic digestion were completed within 25 minutes. The chromatographic separation was completed in 10 minutes with stepwise-gradient elution. Chromatographic peaks interfering with the surrogate peptide and its stable isotope-labeled peptide as an internal standard were not observed from serum digests. The dynamic range of the calibration curve was 2–200 μg mL−1. The intra- and inter-assay accuracies and imprecisions were 92.2–104.5% and less than 10.0%, respectively. Serum nivolumab concentrations ranged from 12–112 μg mL−1 in 14 cancer patients. The measured concentrations in the LC-MS/MS method were strongly correlated with those in the ELISA method (r = 0.92, P < 0.01). In conclusion, a simple and rapid LC-MS/MS method for quantifying serum nivolumab using immobilized tryptic digestion coupled to IgG purification was found to be acceptable for clinical settings.


 Please wait while we load your content...
Please wait while we load your content...