Combination of MALDI-TOF MS and UHPLC-ESI-MS for the characterization of lytic polysaccharide monooxygenase activity†
Abstract
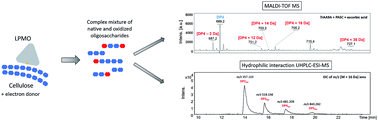
Lytic polysaccharide monooxygenases (LPMOs) are redox enzymes of high biotechnological interest due to their capacity to degrade recalcitrant polysaccharides, such as cellulose, by an oxidative mechanism. The characterization of LPMOs is challenging since they generate a variety of catalytic products which include native oligosaccharides (non-oxidized) and oligosaccharides oxidized at the reducing end (C1 position), the non-reducing end (C4 position), or both ends, with different degrees of polymerization. Moreover, oxidized products exist in equilibrium with their hydrated forms, which further complicates their identification. The lack of commercial analytical standards for all these possible forms of oxidized oligosaccharides and the low concentration of these products make LPMO functional characterization dependent on advanced mass spectrometry techniques capable of identifying the profile of oxidized products. Here, a new approach for the characterization of cellulose-active LPMOs based on the combination of MALDI-TOF MS and hydrophilic interaction UHPLC-ESI-MS was proposed and optimized. The LPMO TrAA9A from Trichoderma reesei was used as a model enzyme to develop and test the MS methods. MALDI-TOF MS and UHPLC-ESI-MS methods were both capable of identifying putative C1, C4 and C1/C4 oxidized cello-oligosaccharides as well as their native counterparts generated after cellulose treatment with LPMO, which allows their utilization to characterize type I (C1-oxidizer), type II (C4-oxidizer) and type III (C1- and C4-oxidizer) LPMOs. Moreover, both methods were complementary since MALDI-TOF MS was capable of detecting oligosaccharides with higher degrees of polymerization (DP3–DP10), while UHPLC-ESI-MS allowed the evaluation of smaller oligosaccharides (DP1–DP5). The combined use of both methods offers a comprehensive description of LPMO catalytic products.


 Please wait while we load your content...
Please wait while we load your content...