An amino functionalized magnetite nanoparticle and ionic liquid based electrochemical sensor for the detection of acetaminophen
Abstract
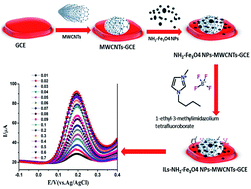
Acetaminophen (AMP) is a pain reliever (painkiller) and a fever reducer. It is also used against arthritis, colds, muscle aches, and headaches worldwide. Because of its increased used in the past several decades, among painkillers, acetaminophen is now present at higher concentrations in pharmaceutical samples, thus necessitating its monitoring. A novel composite electrode has been fabricated by using multi-walled carbon nanotubes (MWCNTs), amino group functionalized magnetite nanoparticles (NH2-Fe3O4NPs) and 1-ethyl-3-methylimidazolium tetrafluoroborate ionic liquid (IL) for the sensitive electrochemical detection of AMP. The synthesized functionalized magnetite nanoparticles were characterized by Fourier transform infrared spectroscopy (FTIR) and the surface morphology and elemental composition of the fabricated sensor were analyzed by transmission electron microscopy (TEM) and energy dispersive X-ray spectroscopy (EDX). In addition, various parameters such as effect of pH, effect of deposition time, effect of sweep rate, and effect of potential, time and interferents were also successfully monitored. The fabricated sensor enhanced the peak currents over 50 times greater than the pure glassy carbon electrode in cyclic voltammetry (CV). Differential pulse voltammetry (DPV) gave a selective determination of AMP without any interference via the standard addition process. The proposed electrochemical sensor showed good concurrence with previously reported methods and was effectively employed for the quantification of acetaminophen (AMP) in various pharmaceutical samples.


 Please wait while we load your content...
Please wait while we load your content...