An octahedral Cu2O@AgNCs substrate in liquid-microextraction coupled chemometric algorithms for SERS sensing of chromium(iii & vi) species†
Abstract
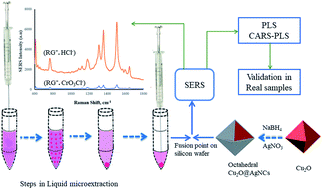
The current study assembles liquid-microextraction, surface enhanced Raman spectroscopy (SERS) and chemometric algorithms in one platform for chromium speciation using octahedral Cu2O@Ag nanocomposites (Cu2O@AgNCs) as a SERS substrate. The reaction between cationic dye rhodamine 6G (RG+) and the chlorochromate anion (CrO3Cl−) at pH < 1.0 has been exploited for chromium determination by the phenomenon of ion association. The conditions of microextraction were carefully established, followed by depositing the sediment organic phase on the ethanolic octahedral Cu2O@AgNCs suspension. The SERS intensity was recorded for the complex ion associate (RG+·CrO3Cl−) versus a reagent blank. Chemometric algorithms such as partial least squares (PLS) and variable selection competitive adaptive reweighted sampling (CARS)-PLS were applied for masking redundancy in the SERS spectra of the complex ion associate (RG+·CrO3Cl−) formed in the sediment phase extract. Substrate characterization using scanning electron microscopy (SEM), transmission electron microscopy (TEM), X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), energy dispersive spectroscopy (EDS), stoichiometry, analytical applications, selectivity and standard method validation was performed. The designed octahedral Cu2O@AgNCs/RG+/HCl probe displayed a detection limit of 0.028 μg L−1 for chromium(VI) under optimized chemometric modeling and microextraction conditions.


 Please wait while we load your content...
Please wait while we load your content...