Detection of degraded, adulterated, and falsified ceftriaxone using paper analytical devices†
Abstract
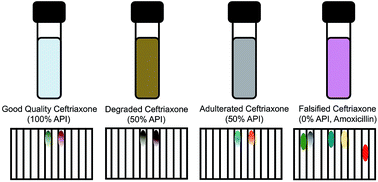
Substandard or falsified versions of ceftriaxone have been found in the supply chains of many countries. Ceftriaxone is an injectable antibiotic often used for treatment when resistance to other antibiotics has developed, so detection of bad quality formulations is of keen interest to the global health community. We found that a paper analytical device (PAD) can detect and differentiate degraded, adulterated, and falsified ceftriaxone injectable formulations. PAD analysis of eighty blinded formulations prepared in lab (20 falsified, 20 thermally degraded, 20 adulterated, and 20 good quality) had a 95% accuracy in classifying the formulations. Forty dosage forms collected in Western Kenya were also assessed, and the PADs correctly identified three of four substandard samples. The sample that was missed had an 87.1% ceftriaxone content, which is just outside the 90–120% pharmacopeia range. The deterioration observed during storage of ceftriaxone solutions at room temperature shows a 2–3 day induction period, followed by rapid auto-catalyzed base degradation that quickly destroys the ceftriaxone. The instability of this drug highlights the need for inexpensive point-of-care testing to monitor ceftriaxone quality.
- This article is part of the themed collection: Bioanalytical sensors for real world applications


 Please wait while we load your content...
Please wait while we load your content...