Reduction of blood volume required to perform paper-based hematocrit assays guided by device design†
Abstract
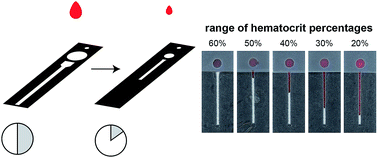
Point-of-care diagnostic tests can provide rapid and accurate information about the health of a patient without relying on the expensive equipment found in centralized laboratories. Not only does the development and application of these assays rely on the means of signal transduction, but also the method by which a sample is procured. In order for point-of-care tests to have real clinical utility, they must use sample types and volumes that are relatively easy to obtain (e.g., fingerstick volumes of blood). In this paper, we demonstrate how the design of a paper-based microfluidic device controls the transport of blood within the device and ultimately influences the development of an assay to measure the hematocrit. We show that, with directed design modifications, altering the dimensions of paper-based devices can greatly reduce the volume of blood required to initiate a hematocrit assay (from 50 μL to only 10 μL) without impacting its analytical performance.


 Please wait while we load your content...
Please wait while we load your content...