Construction of salicylaldehyde analogues as turn-on fluorescence probes and their electronic effect on sensitive and selective detection of As(v) in groundwater†
Abstract
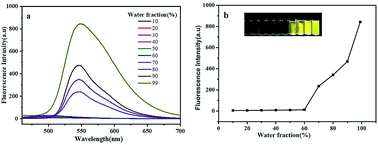
Arsenic contamination in groundwater is a severe and major environmental problem that endangers public health. As such, a sensitive and selective methodology for the detection of this toxic metalloid must be urgently developed. Aldimine condensation reactions of p-phenylenediamine with salicylaldehyde derivatives yielded “turn-on” fluorescence probes (HBHP, HBBP and HMBP) for the detection of As(V) with high sensitivity. Among these, as a typical AIEgen, HBHP exhibits the highest detection sensitivity. The detection limit of HBHP for As(V) was calculated to be 0.88 ppb, which is due to its higher ability to form intermolecular hydrogen bonds to convert twisted intramolecular charge transfer (TICT) into planar intramolecular charge transfer (PICT) and its higher molecular planarity triggered by specific electronic effects. Job's plots and DFT theoretical studies indicate that HBHP binds to As(V) with a stoichiometry of 1 : 2, and the binding constant was determined using the Benesi–Hildebrand equation to be 4.26 × 104 M2. Commonly existing anions and cations in groundwater exhibited no interference with the selective detection of As(V). Spiking and recovery tests of HBHP in the presence of As(V) in actual water samples gave satisfactory experimental results. Our research would provide novel principles of molecular construction and materials for the detection of As(V) in groundwater.


 Please wait while we load your content...
Please wait while we load your content...