Dark-field microscopy for characterization of single molecule dynamics in vitro and in vivo†
Abstract
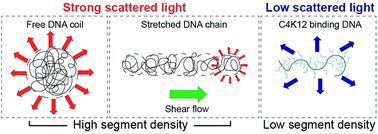
Dark-field microscopy directly detects scattered light from a sample, and therefore requires no fluorescent labeling for single molecule detection. The technique has been extensively used for spectral characterization of nanoscopic particles, but its sensitivity to variations in molecular density has not been explored. 3D chromosome topology plays important roles in regulating life processes. Physical characteristics of DNA (e.g. persistence length) and its interaction with numerous proteins and polyamines determine chromosome and chromatin 3D topology. We herein demonstrate the capacity of dark-field microscopy in real-time monitoring of single DNA molecules' conformation and dynamics. Without fluorescent staining, DNA molecules remain intact in physical characteristics like persistence length, net charges and width of the chain. Exclusion of photobleaching and photocleavage effects allows long-term imaging of bare DNA chains at high ionic strength, in shear flows and in the presence of condensing and stretching agents. Furthermore, our studies reveal that the brightness of single molecules during dark-field imaging is determined by DNA conformation, dark when being stretched and bright upon condensation. The sensitivity of dark-field microscopy to nonuniform distribution of molecular density allows us to study the dynamic activities of the transcriptional factor within the cell membrane. In contrast to a fluorescently tagged antibody, which binds to a specific signaling pathway, variations in molecular density across the nuclear envelope can better represent cellular responses upon stimulation.


 Please wait while we load your content...
Please wait while we load your content...