Compact and efficient gas diffusion electrodes based on nanoporous alumina membranes for microfuel cells and gas sensors†
Abstract
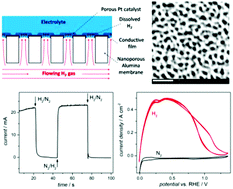
This work describes the fabrication, characterization and evaluation of thin gas diffusion electrodes (GDEs) that are capable of electrocatalyzing the reaction of dissolved gases operating at high diffusion limiting current densities and with fast response times. Nanoporous alumina membranes (NAMs) were used as supports of metal electrocatalysts. NAMs were hydrophobized by silanization and coated on one side with Pt, either over the whole alumina surface by sputtering or just onto the pore openings by local chemical deposition. The Pt-modified NAM-based GDEs were evaluated for hydrogen oxidation. They operated by exposing their coated side to the electrolyte solution and the hydrophobic uncoated side to the flowing gas. Due to the NAM hydrophobicity, flooding of the pores by the electrolyte was diminished, so they were quickly filled by the circulating gas. Simulations of the process on single-pore GDEs showed that the dissolved gas diffused into the solution both radially (ideal pore) and linearly (partially flooded pore), reaching the electrocatalyst film placed right around the pore, which guaranteed a fast mass transport. Hydrogen oxidation operated on these NAM-based GDEs at a steady state with limiting current densities as high as 0.5 A cm−2, which were attained in less than 5 s and were proportional to H2 concentrations over a wide range. Thus, their potential use in microfuel cells and gas sensors was demonstrated.
- This article is part of the themed collection: Versatile Electrochemical Approaches


 Please wait while we load your content...
Please wait while we load your content...