A highly selective ratiometric fluorescent probe based on naphthalimide for detection and imaging of CYP1A1 in living cells and zebrafish†
Abstract
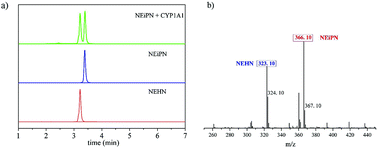
Real-time monitoring of the cytochrome P450 1A1 (CYP1A1) activity in complex biological systems via a practical tool is highly sought after because of its significant role in the metabolism and bioactivation of various xenobiotics. Herein, according to slight differences in the 3D structure and substrate preference between CYP1A1 and its homologous CYP1A2, a series of novel ratiometric fluorescent probes were designed and synthesized using 1,8-naphthalimide because of its trait of naked-eye visualization and ratiometric fluorescence to achieve the detection of CYP1A1 in biological samples. Among these probes, NEiPN showed good water solubility, highly isoform selectivity and great sensitivity (LOD = 0.04874 nM) for CYP1A1 under simulated physiological conditions, which makes it favorable for monitoring CYP1A1 in vivo. Remarkably, NEiPN exhibited excellent reproducibility when it was used to detect the CYP1A1 content in human liver microsomes, which indicated that it has a great potential for quantifying the CYP1A1 content in real biological samples. Furthermore, NEiPN showed relatively low cytotoxicity and has been successfully applied in biological imaging in living cells and zebrafish. These findings indicate that NEiPN is capable of real-time monitoring of the activity of endogenous CYP1A1, which could provide support for CYP1A1-associated pathological processes.


 Please wait while we load your content...
Please wait while we load your content...