Metabolomics for improved treatment monitoring of phenylketonuria: urinary biomarkers for non-invasive assessment of dietary adherence and nutritional deficiencies†
Abstract
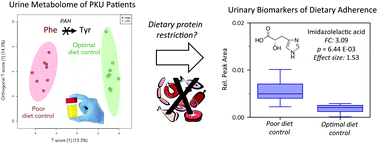
Management of phenylketonuria (PKU) requires lifelong restriction of phenylalanine (Phe) intake using specialized medical foods to prevent neurocognitive impairment in affected patients. However, dietary adherence is challenging to maintain while ensuring adequate nutrition, which can lead to sub-optimal clinical outcomes. Metabolomics offers a systematic approach to identify new biomarkers of disease progression in PKU when using urine as a surrogate for blood specimens that is more accurate than self-reported diet records. Herein, the plasma and urine metabolome of a cohort of classic PKU patients (median age = 11 years; n = 22) mainly prescribed (78%) a Phe-restricted diet were characterized using multisegment injection-capillary electrophoresis-mass spectrometry (MSI-CE-MS). Overall, there was good mutual agreement between plasma Phe and tyrosine (Tyr) concentrations measured from PKU patients when using an amino acid analyzer based on UPLC-UV as compared to MSI-CE-MS with a mean bias of 12% (n = 82). Longitudinal measurements of recently diagnosed PKU infants (n = 3) revealed good long-term regulation of blood Phe with dietary management, and only occasional episodes exceeding the recommended therapeutic range (>360 μM) unlike older PKU patients. Plasma metabolomic studies demonstrated that non-adherent PKU patients had lower circulating concentrations of Tyr, arginine, 2-aminobutyric acid, and propionylcarnitine (q < 0.05, FDR) that were inversely correlated to Phe (r ≈ −0.600 to −0.830). Nontargeted metabolite profiling also revealed urinary biomarkers associated with poor dietary adherence among PKU patients, including elevated concentrations of catabolites indicative of Phe intoxication (e.g., phenylpyruvic acid, phenylacetylglutamine, hydroxyphenylacetic acid). Additionally, PKU patients with poor blood Phe control had lower excretion of urinary compounds derived from co-metabolism of Tyr due to microbiota activity (e.g., cresol sulfate, phenylsulfate), as well as several metabolites associated with inadequate nutrient intake, including low carnitine and B vitamin status (e.g., folic acid, vitamin B12). Interestingly, an unknown urinary metabolite was strongly correlated with Phe excretion in PKU patients (r = 0.861), which was subsequently identified as imidazole lactic acid when using high resolution MS/MS. Overall, urine profiling offers a non-invasive approach for better treatment monitoring of individual PKU patients, which can also guide the design of novel therapies that improve adherence to Phe-restricted diets without acquired nutritional deficiencies.
- This article is part of the themed collection: Bioanalytical tools for enabling precision medicine


 Please wait while we load your content...
Please wait while we load your content...