Sensitive detection of caspase-3 enzymatic activities and inhibitor screening by mass spectrometry with dual maleimide labelling quantitation†
Abstract
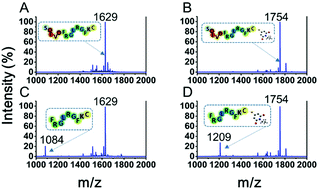
There is a great need to develop sensitive and specific methods for quantitative analysis of caspase-3 activities in cell apoptosis. Herein, we report a new method for sensitive detection of caspase-3 enzyme activities and inhibitor screening based on dual maleimide (DuMal) labeling quantitation and matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS). Evaluation of caspase-3 activities is performed using MS analysis of the enzymatic product of the peptide probe, which fuses a caspase-3 cleavable peptide segment (DEVD) and a quantifiable “ID tag” (a peptide segment of FRGLRGFKC labeled by maleimide). The DuMal labeling technique features non-isotopic tagging, rapid reactions, and reproducible quantitation. We have achieved quantitative analysis of the enzyme activities with a limit of detection (LOD) and limit of quantitation (LOQ) of caspase-3 down to 0.11 nM and 0.29 nM respectively and a proof-of-concept demonstration of its inhibitor screening. Our method has further been tested for caspase-3 activities in a Parkinson's disease cellular model, suggesting a useful tool for protease activity research.


 Please wait while we load your content...
Please wait while we load your content...