Engineering anisotropic cardiac monolayers on microelectrode arrays for non-invasive analyses of electrophysiological properties†
Abstract
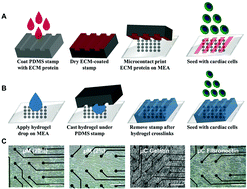
A standard culture of cardiac cells as unorganized monolayers on tissue culture plastic or glass does not recapitulate the architectural or the mechanical properties of native myocardium. We investigated the physical and protein cues from the extracellular matrix to engineer anisotropic cardiac tissues as highly aligned monolayers on top of the microelectrode array (MEA). The MEA platform allows non-invasive measurement of beating rate and conduction velocity. The effect of different extracellular proteins was tested by using the most common extracellular matrix proteins in the heart, fibronectin and gelatin, after aligning myocytes using a microcontact (μC) printing technique. Both proteins showed similar electrophysiological results before the monolayer began to delaminate after the sixth day of culture. Additionally, there were no significant differences on day 4 between the two microcontact printed proteins in terms of sarcomere alignment and gap junction expression. To test the effect of substrate stiffness, a micromolded (μM) gelatin hydrogel was fabricated in different concentrations (20% and 2%), corresponding to the elastic moduli of approximately 33 kPa and 0.7 kPa, respectively, to cover both spectra of the in vivo range of myocardium. Cardiac monolayers under micromolded conditions beat in a much more synchronized fashion, and exhibited conduction velocity that was close to the physiological value. Both concentrations of gelatin hydrogel conditions yielded similar sarcomere alignment and gap junction expression on day 4 of culture. Ultimately, the 3D micromolded gelatin hydrogel that recapitulated myocardial stiffness improved the synchronicity and conduction velocity of neonatal rat ventricular myocytes (NRVM) without any stimulation. Identifying such microenvironmental factors will lead to future efforts to design heart on a chip platforms that mimic in vivo environment and predict potential cardiotoxicity when testing new drugs.
- This article is part of the themed collection: Bioanalytical tools for enabling precision medicine


 Please wait while we load your content...
Please wait while we load your content...