Linker-free antibody conjugation for sensitive hydrogel microparticle-based multiplex immunoassay†
Abstract
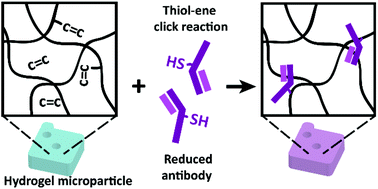
Multiplex immunoassay, or the simultaneous detection of multiple proteins in a single sample, is expected to enable a new level of protein analysis across diverse disciplines, such as medical diagnostics and biomarker discovery. A bead-based assay using graphically encoded hydrogel microparticles synthesized using stop flow lithography has been a promising platform because of its high multiplex capacity and its superior sensitivity and dynamic range compared to the enzyme-linked immunosorbent assay (ELISA). The functionalization of these particles has been dependent on the use of a heterobifunctional linker to conjugate the capture antibodies on the hydrogel. However, the linker chemistry, which is based on linking the primary amine groups of antibodies with acrylate functional groups on the hydrogel monomer, is vulnerable to hydrolysis in aqueous conditions and can potentially damage the antigen binding region of the antibody. In this work, we introduce a new antibody conjugation method that avoids the use of the linker and further enhances the sensitivity of hydrogel microparticle-based immunoassays. Disulfide bonds in antibodies are reduced to liberate free thiols, which can directly bond with the double bonds remaining in the hydrogel after particle synthesis. We characterize the optimal reduction of antibodies for producing the highest detection signal and demonstrate an average two-fold improvement in sensitivity compared to the linker-dependent antibody conjugation method. Lastly, we validate the accuracy and specificity of the multiplex assays with particles conjugated with antibodies using the linker-free method.


 Please wait while we load your content...
Please wait while we load your content...