Charging and supercharging of proteins for mass spectrometry: recent insights into the mechanisms of electrospray ionization
Abstract
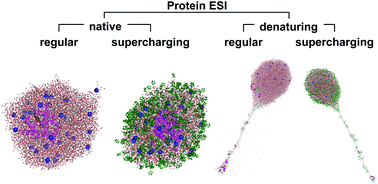
Electrospray ionization (ESI) is an essential technique for transferring proteins from solution into the gas phase for mass spectrometry and ion mobility spectrometry. The mechanisms whereby [M + zH]z+ protein ions are released from charged nanodroplets during ESI have been controversial for many years. Here we discuss recent computational and experimental studies that have shed light on many of the mysteries in this area. Four types of protein ESI experiments can be distinguished, each of which appears to be associated with a specific mechanism. (i) Native ESI proceeds according to the charged residue model (CRM) that entails droplet evaporation to dryness, generating compact protein ions in low charge states. (ii) Native ESI supercharging is also a CRM process, but the dried-out proteins accumulate additional charge because supercharging agents such as sulfolane interfere with the ejection of small ions (Na+, NH4+, etc.) from the shrinking droplets. (iii) Denaturing ESI follows the chain ejection model (CEM), where protein ions are gradually expelled from the droplet surface. H+ equilibration between the droplets and the protruding chains culminates in highly charged gaseous proteins, analogous to the collision-induced dissociation of multi-protein complexes. (iv) Denatured ESI supercharging also generates protein ions via the CEM. Supercharging agents stabilize protonated sites on the protein tail via charge–dipole interactions, causing the chain to acquire additional charge. There will likely be scenarios that fall outside of these four models, but it appears that the framework outlined here covers most of the experimentally relevant conditions.


 Please wait while we load your content...
Please wait while we load your content...